Educational Effect of Microcomputer-Aided Learning on
Practical Experiments in Chemistry Curriculum
Tadayosi Yoshimura*1, Hideo Sakagami*1, Yoshihiro Fujikami*2
and Brian T. Newbold*3
 Return
Return
Introduction
The chemistry curriculum should have the minimum numbers of fundamental lectures and experimental lessons for schooling in Japan[l,2,3,4,5]. In fact, the more extensive the experimental experience, the more effective the learning will be. Since chemistry is an experimental science, it is very important that students have hands-on experience in dealing with chemical substances.
A experiment is very important in a chemistry class. Some students may think that hands-on experience learning is easy for cutting corners ! A student may have to receive training before a class due to the circumstances of the chemistry curriculum. Despite this, the student might neglect learning before the experimental class.
In a limited curriculum, the way to increase the educational effect of experience learning has been dealt with educationally. A new approach to teaching chemistry has to be complementary to the educational method used (textbook and blackboard). Computer-aided instruction (CAI) can offer a solution to this problem, and also encourage chemistry teaching. Therefore, we have tried to let students do some learning before the practical training by the use of the microcomputer.
Microcomputer-aided learning (CAL) has been put into effect via testing in chemistry teaching on the spot. The students learn about experimental contents by themselves before starting the actual practical work.
Preparation of CAL software
We have been in charge of physical chemistry experiments for a chemistry class. Formerly, however, we did not have CAL software for beforehand learning for physical chemistry experiments. Computer input can be used as a learning tool. We impose learning before the experimental training and examine the beforehand learning effect on education. There are l8 themes, as shown in Table l.
No. chapter title
1 neutralization heat between acid and base solutions
2 dilution heat of dimethylformamide into water
3 molecular weight of acetone by Victor-Meyer method
4 molecular weight of nitrobenzene by steam distillation method
5 molecular weight of naphthalene by depression of freezing point
method
6 solubility of benzoic acid and the dissolution heat
7 partition coefficient between aqueous and tetrachloromethane
phases
8 ionization constant of acetic acid
9 physical properties (viscosity, density etc.) of ethanol and water
solution
10 hydrolysis of ethyl acetate in acidic solution
11 hydrolysis of ethyl acetate in basic solution
12 inversion reaction of sucrose solution
13 adsorption of acetic acid on activated charcoal
14 point of zero charge of metal oxide colloid
15 electromotive force of concentration cell
16 decomposition voltage of electrolyte solutions
17 transport number of HCl solution
18 potential difference titration of neutralization
The student must understand the experimental operation necessary for training beforehand in each theme. In order to develop CAL, we have prepared three softwares: Japanese word processor, graphic processor, and building shell system[4,5]. Incidentally, the CAL software is made perfectly and easily for the desired physical chemistry experiment courseware. These softwares cover the l8 chapters mentioned above and each one consists of four sections:
l) theory and principle,
2) experimental (apparatus and device),
3) experimental operation,
4) result and discussion.
Each section has one or two questions as a drill. The student not only learns physical chemistry unilaterally, but also answers a question. He/she will understand an experimental subject through beforehand learning. Figures l, 2, 3, and 4 show samples of the CAL display of chapter 5 (molecular weight of naphthalene by depression of freezing point method). These hardcopies are made by photocopying, so they show up in black and white. In reality, the displays are in beautiful color. This CAL software is made up of two frames: explanation frame and drill frame. For example, the display in Figure l is section 2, which illustrates the diagrammatic representation of a Beckmann thermometer operation. Figure 2 shows an example of a drill frame. Question 5.2 of Figure 2 is the key point for use of the thermometer. Figure 3 shows an example of the experimental operation. The experimental result is explained in a graphic way as seen in Figure 4.
These CAL softwares for physical chemistry experiments are now distributed free to members of the Chemical Software Society of Japan[6]. They are available any time and are very useful for individual learning.
Educational Effect of CAL
In order to investigate the educational effect of CAL learning on the chemistry experimental lesson, we examined the training impact on chemistry students with or without beforehand CAL learning. The students were from our College. The CAL softwares were prepared for half of the l8 themes so as to investigate two cases with CAL or textbook. The CAL softwares concerned chapters l to 9 (Table l). In the case without CAL software the students must learn beforehand by textbook. Figure 5 shows the flow scheme for a practical experiment with beforehand learning by textbook or CAL. We have two types of student's reports, which are from beforehand learning by textbook(report A) and CAL(report B). The students must understand the experimental operation necessary for training beforehand in each theme (physical chemistry experiment). However, there are some students who would set up beforehand learning for cutting corners! Lazy students ignore beforehand learning for practical experiments.
The 67 students involved were divided into two groups on the basis of their final score. Group A consisted of 3l students who got less marks than the mean score. Group B was made up of 36 students who had better marks than the mean score.
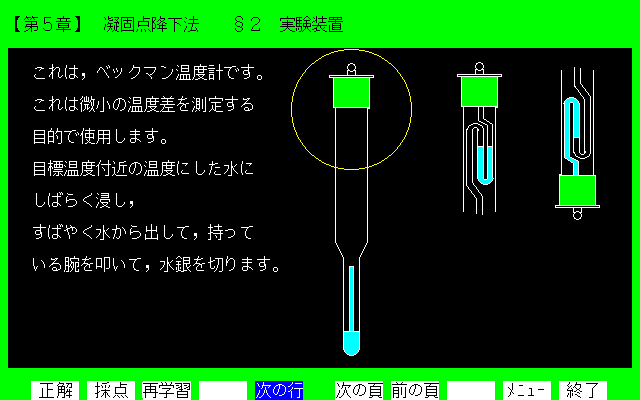
Figure 1. Display of section 2 in chapter 5.
[Explanation] This diagrammatic representation is a Beckmann thermometer operation. The thermometer has to be used carefully when measuring minute change of temperature in a calorimeter box.

Figure 2. Display of drill frame in chapter 5.
[Explanation] This drill frame (question 5.2) is for operating a Beckmann thermometer. The key point is selected exactly among three operation terms.
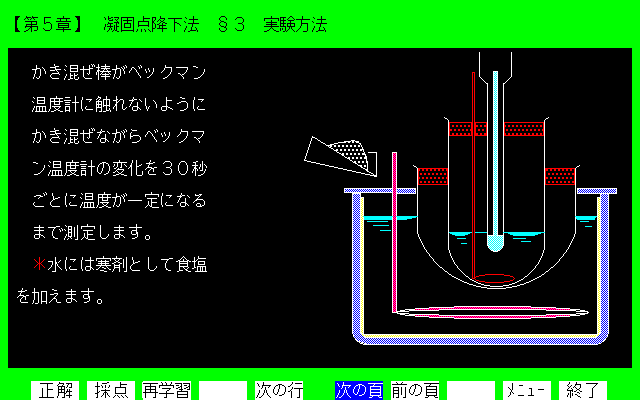
Figure 3. Display of section 3 in chapter 5.
[Explanation] This diagrammatic representation is the experimental operation of depression of freezing point method. The refrigerants are ice and salt.
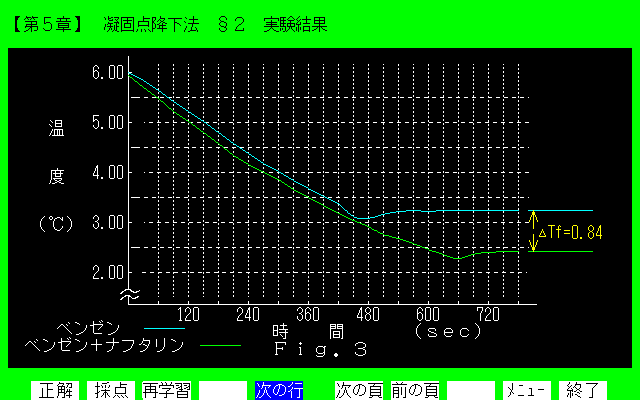
Figure 4. Display of section 4 in chapter 5.
[Explanation] The experimental result is explained in a graphic way. The depression degree .Delta.Tf is determined from this figure.
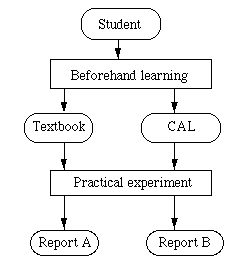
Figure 5. Flow scheme for practical experiment with beforehand learning by texbook (report A) or CAL (report B).
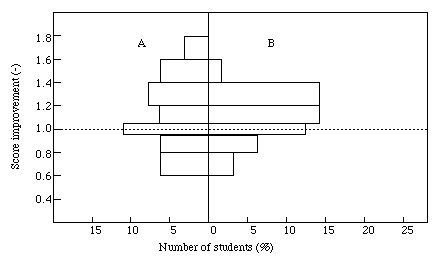
Figure 6. Histogram of the score improvement with or without the CAL softwares.
A : students obtained less marks than the mean score, B : students had better marks than the mean score.
In order to study the educational effect with or without CAL software, we have examined the improvement rate for the student's score. The result is shown in Figure 6. A vertical line in the histogram is a score improvement with or without CAL softwares, which is defined as the score value with CAL software divided by that without CAL software. Thus, if a student gets 8 points per l0 full mark with software and obtains 6 points without software, the score improvement is l.3. The value of the histogram means the % number of students. The left side of the histogram describes the score improvement of students who got less marks and the right side that of those who had better marks than the mean score. It is found that the CAL effect is weak for the lazy student who does not intend to run experimental training, though the score improvement is taken in general from group A in Figure 6. Compared with group A, a student in group B gets better marks with CAL software than one without it, and the score improvement rate concentrates on a histogram of l.0 to l.4.
For the purpose of the beforehand CAL learning effect, we have examined the weight rate of the learning for the student's score. The result obtained is given in Figure 7. A vertical line in the histogram is the weight rate of CAL learning, which is defined as the value that is obtained when the score for CAL learning is divided by the final score. Thus, if a student gets 9 points per l0 full mark from beforehand learning and has 7 points as his/her final mark, the weight rate is l.3. The value of the histogram means the % number of students. The left and right sides of the histogram have the same meanings as those shown in Figure 6. It was found that the beforehand learning effect is weak for lazy students. Compared with group A, a student in group B would get good marks, and the weight rate focusses on a histogram of l.0 to l.2.
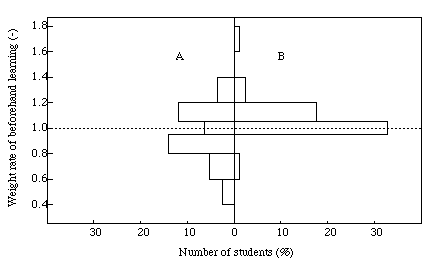
Figure 7. Histogram of the weight rate of beforehand learning on students' scores.
A : students obtained less marks than the mean score, B : students achieved better marks than the mean score.
Conclusion
Chemistry teaching should be advantageously innovated by the use of the microcomputer in association with the practical experiment. Microcomputer software for CAL learning can be used to run beforehand learning for
practical experiments in chemistry. Depending on the nature of the student, it is found that beforehand learning(CAL) for experimental training is educationally effective.
Acknowledgments
The authors wish to express their thanks to Professor John T. Shimozawa of Saitama University, and President Shin-ichi Sasaki of Toyohashi University of Technology for valuable advice and discussions throughout this work.
References
1) J. T. Shimozawa: J. Sci. Educ. Jpn., 8, 131-138(1984).
2) S. Sasaki: Widening the Scope of Chemistry, IUPAC, Blackwell Scientific
Publications (1987) 11.
3) T. Yoshimura: J. Chem. Educ. Jpn., 35, 572-575(1987).
4) T. Yoshimura: J. Chem. Software, 1, 1-14(1992).
5) T. Yoshimura, Y. Fujikami: J. Chem. Software, 1, 89-98(1992).
6) T. Yoshimura(ed.): Annual Report of Chemical Softwares, The Chemical Software Society of Japan (1992).
 Return
Return

 Return
Return
 Return
Return