Two Novel Software Programmes for Assisted Learning - Oxidation Numbers and Chemical Nomenclature
Paul LEBLANC and Brian T. NEWBOLD
 Return
Return
1 Introduction
The use of computer assistance in teaching (CAT) is nothing new. Computer-aided teaching (CAT) has been the logical successor of various teaching endeavours, namely programmed teaching, programmed learning, the various uses of audio-visual tools that were quite popular until the seventies or so. Since then, and at this particular point in time, the use of computer aided instruction (CAI) and computer-aided learning (CAL) is also not new. The advantages and shortcomings of their use are quite evident to those who commonly make use of the various software packages now available. Computers are nevertheless prevalent in the teaching and learning environment of schools, all the way from kindergarten to university. Such conditions foster the use and constant development of various software programmes to enhance the quality of communication between professors and students and of learning and application on the part of students themselves.
The two interactive software programmes presented here fall in the category of computer aided-learning (CAL) rather than in other categories of teaching-related programmes. Both programmes were developed by one of the authors (P.L) at l'Universite de Moncton, for a beginning chemistry course, the intent of which is to streamline students with little or no previous preparation in this field and to bring them to a second semester level in the first year of a four year baccalaureate in the sciences. The topics covered include a theory summary in each case as well as a variety of interactive exercises bearing upon Oxidation numbers and general Inorganic Chemical Nomenclature. Both of these subjects are commonly discussed in any introductory general chemistry course, both at the High School and College levels.[1 - 3]
As alluded to above, students lacking the two preparatory courses in chemistry offered in the Province of New Brunswick's high schools, must follow this university's introductory course CHIM1014 so as to acquire the skills intended by the regular CHIM1013 (for those students with the necessary preparation) in order to enter the subsequent CHIM1023 and to complete the First year requirements in chemistry. The course is given in French. In a normal term, between 60 and 100 students, with varying degrees of chemical knowledge, follow CHIM1014. As French is the language of instruction at l'Universite de Moncton, it is for that reason that the original versions of both programmes were written in that language. An English translation has now been completed for the Oxidation Numbers programme while one for the nomenclature programme is intended.
The programmes that will be described have been used by a large number of students, especially those who wish to perfect their theoretical notions with the help of supplementary exercises. The attraction of computer assistance in their learning process contributes to the willingness to use this type of software. The modules "Oxidation-Reduction" and "Inorganic Chemical Nomenclature" as well as other programmes available in the University's Computer Teaching and Learning Centre help to satisfy this need for further personal work.
2 Oxidation-Reduction
Having personally taught oxidation-numbers for several years, we saw this topic as perhaps the type of module one could try to develop with the use of Authorware as an authoring language. Built-in functions enable non-programmers to produce usable and interactive software which can be easily put to profit by interested students.
Although the final product is extremely easy to use, the learning curve of Authorware is somewhat steep, and one has to devote a good deal of time in order to obtain suitable results. The effort, nevertheless, is worthwhile and student satisfaction as well as their positive comments are always gratifying.
This module was the first to come to fruition. It was subsequently reviewed and augmented and improved in its functionality. As said earlier, an English translation was realized in order to reach a number of students far exceeding the clientel of francophone students for which the module was first intended.
Professor Jim Spain of Clemson University, a well-known authority and contributor on the scene of computer-assisted learning, was one of the first experts in this field to try out the original French version, "Oxydo-reduction". His comments were most encouraging and flattering . One of us (P.L) was invited to present (although the translation had not yet been done) the programme at the "Electronic Homework" workshop held at the Michigan Technical University, Houghton, Michigan in July, 1996. The interest which ensued from this initial presentation spurred further development of this type of software module. This led to the development of the nomenclature module. A module on significant figures is now foreseen as well as another one on nomenclature.
Both of the existing modules have a similar structure. Each begins with a short animation bearing on the subject, the requirements and what the student should have at hand and do during the exercises. This is followed by a main menu (Figure 1). Heading this menu is an invitation to click on button 1, "Theoretical Notions". This item contains several notions and sub-notions dealing with oxidation numbers, with the rules to derive these numbers and the calculations that apply in various cases along with some examples. All this, is quite succinct and needs only two or three screens.
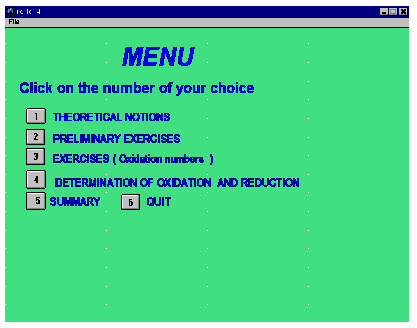
Figure 1. Main menu
Concluding this initial presentation, one is called upon to decide upon a relatively simple case. Each notion is then opened by a simple click of the mouse on several buttons. All in all, this initial treatment constitutes a very small part of the entire programme. Also, it is only a reminder of notions normally covered previously by the instructor and students. In fact, neither of the two modules described herein pretends to be complete as a learning tool. To maximise his or her profit from this software, the student must indeed master beforehand the concepts and knowledge which go with the exercises. Finally, the sub-menu "Theoretical Notions " represents the least interactive component of the entire module. This also applies to the Nomenclature module.
3 More specific aspects and content
As the English and French versions are identical in content and structure, what is said of one version applies to the other.
On the Main Menu, the user can select exercises that go from simple to more complex. Immediately after the "Notions" button, the menu offers some "Preliminary exercises". With these, a sub choice is again offered: "Fundamental" and, afterwards, "More advanced" questions are proposed in the menu.
All this part and the next are totally interactive. The user is clearly instructed to execute certain operations either by the use of the keyboard or by mouse clicks. Thus, one is invited to enter certain values along with the proper signs of the oxidation numbers or by clicking on buttons displaying selected values of which only one is correct. At times, not only values are to be entered, but words also. Authorware provides "hot spots" for entering key terms. True or false buttons, allow the proper selection in multiple choice questions and timed delays can be given for a desired answer.
The accompanying Figure 2 is an illustration of a timed question. In this particular case, to avoid undue frustration, a second chance is given. This should always be the case. It often happens that the length of the question and the time taken to read it do not allow for a proper decision by the student. Nevertheless, if one misses the first chance, the fact that the time is up, evidenced by the blackened clock, prompts one to come to grips more consciously with the request and then have a fair chance at succeeding upon the second try. In itself, a timed question is fine for those who are rapid readers and executants. However consideration has to be given to the legion of those "middle of the road " students, hence the second try. If the timed questions serve as a wake up call, then their purpose is worthwhile.
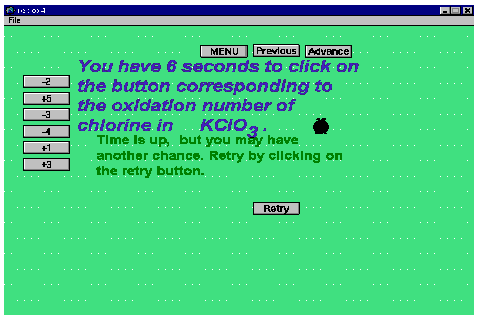
Figure 2. Illustration of a timed question
In all cases, varied oxidation number operations are commonplace by clicking on buttons 2 and 3 of the Main Menu. Students must realize the importance of oxidation numbers, in spite of their artificial character, for balancing redox equations, and to a lesser degree, when considering polarities and induced electronic effects.
The electron transfer method, more so than the half-reaction method, relies heavily on the correct usage of oxidation numbers. One has to know, or at least be able to calculate the proper values. It is with this in mind that the various exercises are proposed in this software.
Easy to use, congenial, " user friendly ", even to the computer uninitiated, this software gives an immediate and respectful reply to the learner. Often, a hint or a prompt will orient the user towards a sensible response. In the case of a wrong reply, or after the last chance, the correct answer appears on the screen. In most cases, a supplementary pedagogical commentary accompanies the answer. Care has been taken to avoid all derision, and comments are meant to provide encouragement.
Each screen keeps a certain general and common appearance. For each given question, the navigation buttons always show in the same area, i.e. the upper right-hand corner. These buttons allow for easy navigation throughout the programme. Three such buttons constitute the essential upper levels of navigation. With these buttons, one can go ahead, back up or go to the Main Menu. Double clicking on the advance button does not automatically mean a skipped question. This is so because each question asks for confirmation in order to proceed to the next. Should the need arise to go rapidly to the end of a series of questions, the Menu option is undoubtedly the best, rather than having to click through all the questions.
In the French version, the imperative polite form (second person plural) is constantly used rather than the imperative (second person singular). Of course, this is a non-issue in the English version. Likewise, even though a certain amount of "lightness" or humour is used in the answers, every effort was made to maintain an attitude of respect towards the learner.
Screen background is constant. A restful pale green is the usual backing upon which the questions are posed. However, to avoid monotony and with the visual diversity sought by most of today's students, the questions and the general appearance of frames or boxes or highlighted areas take on different looks from one screen to another. Also, the style of interaction varies throughout the exercises. The same goes for colour, size and fonts as applied to each scenario. All this affords the type of variety that was shown to be appreciated by the users.
In a general way, throughout this software, the answers or the invitation to retry are given in the lower left portion of the screen. In the case of a wrong answer, the computer reply appears in red whereas congratulatory remarks for right answers always appear in blue within the same area.
Button 3 requests that the student perform calculations, generally so as to enter all the algebraic values of the oxidation numbers of each element in the formula of a given compound. Obviously, only the correct combination of numbers and signs is accepted. As students are prone to distraction, they must work with a good degree of concentration in order to succeed in these exercises. Many entries are made via the keyboard, but for the sake of diversity, other means of input are also solicited, whether it be by clicking on a correct sequence of buttons, on true or false choices or others. Again, the graphic layout varies from one question to another.
With button 4, the student is confronted with another type of question. In this case, he or she must determine the oxidizing agent or the reducing agent as the case may be. This type of question represents a higher degree of difficulty. Beginning students often find difficulty in identifying oxidizing and reducing agents, as well as oxidized and reduced substances.
These exercises are illustrated by redox reactions taken from well-known every day situations. These vary from the simple manifestation of corrosion of a metal, the colour change of the dichromate ion in the presence of ethanol in the breathalyser test to the propulsion reaction found on the space shuttle, Endeavour. In these cases, one indicates his or her choice by clicking directly on the elements or compounds that are linked by a "hot spot" to the correspondingly correct answer. In some cases, certain answers are accompanied by a few words of complementary explanation or hints to further encourage the search for a better answer.
Button 5 provides a brief sequence of screens that summarize the notions that were covered in this learning software, whereas button 6 is used to quit the programme. When the latter is the chosen option, a note of thanks is expressed as well as an invitation for comments.
Although this "Oxidation-Reduction" programme does not provide for random questions nor furnish the results of the student's success and mistakes, the users have shown consistent satisfaction with this software in order to verify their knowledge, their ability to apply it and to gain more confidence in their relevant skills.
4 Inorganic Chemical Nomenclature
As this software programme is part of an intended series, it is no surprise to find similarities in the structure and the appearance of this second learning module. Likewise, the same pedagogy applies and underlies its conception and realization.
The main menu does however show an additional button, namely that which leads to the periodic table. This table shows only the chemical symbols and is structured according to the roman numbers at the head of each group or family. This is preferred over the 1 to 18 order which doesn't give the student the opportunity to quickly realize the charge or the oxidation number of an element within a compound. Such a consideration comes into play where nomenclature is concerned. The chemical names of the elements were omitted in order to stress the importance there is for students to personally know the names of the more frequently used elements. Finally, the periodic table remains accessible for any exercise, although one must return to the main menu. The return to the problem is not automatic. The user would gain from having his or her own table at hand.
5 Structure and content
As in the foregoing learning programme, button 1 of the Main Menu offers a range of general theoretical notions. These are presented with the same approach as that used in the teaching of chemical nomenclature at l'Universite de Moncton.
Rather than learning tables of ions, as is the case in many books, even the most recent, [4 - 6] six hydracids along with their gaseous homologues are given in a table that must be known. Once this is mastered, the student is required to underscore the negative ion (anion) on the formula itself. The charge of the anion corresponds to the number of associated hydrogens. For instance, the sulfide ion will always be 2- because of its association with two hydrogen atoms in H2S.
The same procedure is applied to approximately a dozen common oxyacids. Priority is given to learning those oxyacids ending in "..ic". Less time can then be devoted to memorizing the formulas of the oxyacids ending in "..ous" or qualified by "hypo..ous" and "per..ic" nomenclature.
One will immediately note here that this treatment of the nomenclature, at least to this point, dwells on a classical approach, i.e. not stressing the more descriptive, yet logical and easy system proposed by IUPAC. This approach also recommends the Stock system of nomenclature [4] for ions having multiple charges, such as Iron(II) and Iron(III). Although the Stock system is well taught, the -ous and -ic names are also presented. This approach is somewhat justified by their still common use in the literature, the cataloging and labelling of inorganic chemicals. The IUPAC system, (such as Iron(II)trioxonitrate, etc.) although taught in the classroom, is not adequately covered in the present set of exercises.
Upon mastering the two tables of hydracids and oxyacids, in fact a relatively low number of formulas, and with the immediate and automatic knowledge of the charge associated with each anion, it is estimated that the student already knows 80-90% of the nomenclature. At this point, one can write out the correct formulas of an enormous number of salts. Obviously, the finer points of combining the right proportions of cations and anions only come with exercise and practice. This is the object and scope of courses and also of this particular software programme.
Other buttons lead to more classes of compounds, such as the hydrides, hydroxides, oxides and other non-metallic compounds.
From button 2, the level of interaction increases as the user can begin applying his or her knowledge to direct applications with constant feedback. In most cases, entries are either done with the use of the keyboard or by clicking various buttons offering various choices or decisions, true or false statements. There are some timed questions, just as there were in the Oxidation-Reduction programme. However, there are new modes of interaction in this programme as compared to the previous one. Motion was introduced in some questions. For example, one must drag (Figure 3) ions or fragments of formulas to a predetermined area so as to make up a correct formula or accomplish some other task.
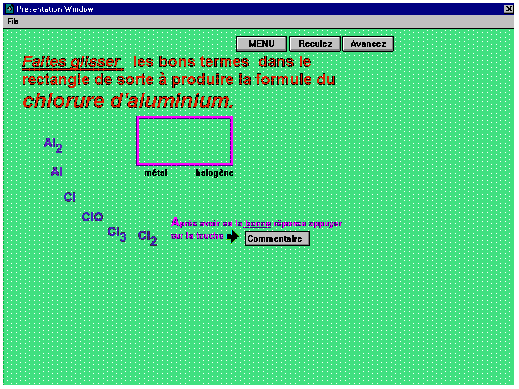
Figure 3. Illustration of motion
The element or ion that was selected will not stay in the designated area if it was wrongly chosen by the student. Rather, it will return to its original place and the screen will show a useful comment or an invitation to try again. The last exercise of the series under button 3 displays the maximum of movement wherein one must click on a particular compound amongst others also in motion. This exercise necessarily requires a speedy identification of the compound combined with a good degree of coordination. In any case, a certain game element underlies the operation.
Unfortunately, Authorware Academic is somewhat limited in the number of icons (built-in functions) that allow for the various programming operations. As motion requires not a few of these icons, the number of exercises of this nature had to be limited.
All in all, it is felt that a good selection of situations is presented. These propose a sufficient number of interactions from the identification of simple and polyatomic ions to the correct formulation of given compounds ( keyboard entries), multiple choices by the correct clicking on proposed buttons or true or false buttons, etc. So as to avoid the sensation of being trapped by timed questions, the student is given a second chance, should she or he overrun the alloted time. The "movement" questions offer more of a game element and are much less "menacing" to the student. Due to their demand on the programme and the programming limitations, their use was somewhat reduced.
6 Requirements
As the two above-mentioned programmes were initially developed with the use of a 486 MHz computer and the Window 3.1 environment, these programmes run on any computer (286 MHz and above) with Windows 3.X, 95 or 98. Unfortunately, being compiled in 16 bit, they cannot operate under NT or Windows 2000 at the present time.
However, with Internet Explorer, one can freely access the download site http://www.sciences.umoncton.ca/ftp_serv/chim/paul/null.htm , and execute directly from the server by using InternetExplorer 5. Such a feature does not appear to be present in Netscape Communicator. The programmes are packaged as self-executable, i.e. they are in the *.exe form on diskettes or hard disks. For optimal speed, one simply copies the programmes to a hard disk (from diskettes or by downloading) and launches the programme by double clicking on its name.
7 Conclusion
Users of either or both of the above-mentioned software have commented very favorably on their usefullness and ease of use. These have been professors present at the "Electronic Howework Workshop" in Michigan as well as peers and students. The only unfavorable comment from one particular student concerned the choice of colour in one screen, namely the exercise on ammonium nitrate. Many have downloaded the programmes since they were made available at P.LeBlanc's homepage[7]. Ideally, one would hope for interactivity on the Web through the use of ShockwaveTM. Version 2.0 of Authorware does not accomodate Shockwave. Version 3.5 (Academic), which can accomodate Shockwave was tried, but then, another limitation made the porting over impractical, if not impossible. Version 3.5 is limited to fewer icons (350) as compared to the 500 allowed in Version 2.0. As both software programmes are in the vicinity of 500 icons, the only practical way for free distribution is by making them available to downloading.
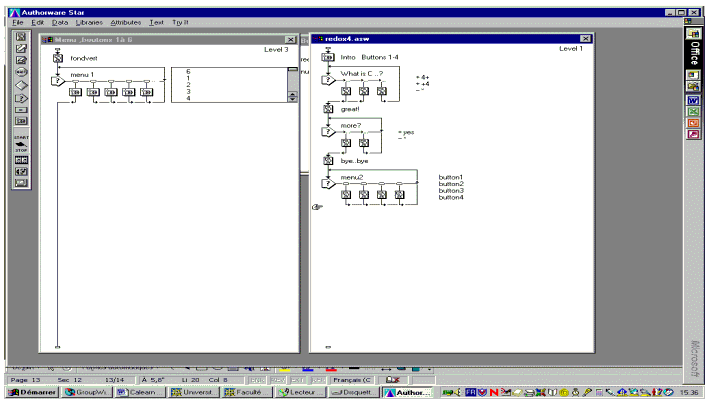
Annex : Typical flow chart in Authorware
References
[ 1] Radel, S.R., Navidi, M. H., Chemistry, West (1994), p.69 sq.
[ 2] Ebbing, D.R., Wrighton, M.S., General Chemistry, Houghton-Mifflin Co. (1990), p.50 sq.
[ 3] Umland, J.B., Bellama J.M., General Chemistry, 2nd ed., West (1996), p.A-1.
[ 4] Burns, R.A., Fundamentals of Chemistry, 3rd edition, Prentice-Hall (1999), p.160.
[ 5] Stoker, H.S., Introduction to Chemical Principles, 6th edition, Prentice-Hall (1999).
[ 6] Corwin C.H., Introductory Chemistry, Concepts & Connections, 2nd edition, Prentice-Hall (1999), p.213-214.
[ 7] WWW site of P.LeBlanc:
http://www.sciences.umoncton.ca/ftp_serv/chim/paul/null.htm
 Return
Return

 Return
Return


 Return
Return