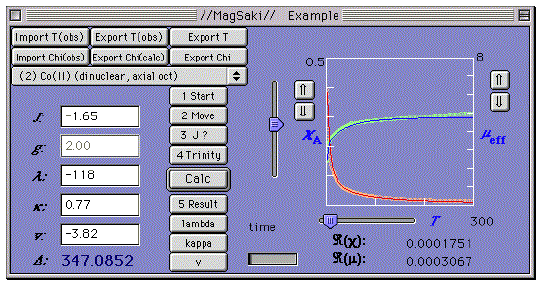
Figure 1. Main window of MagSaki.
 Return
Return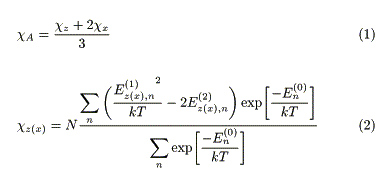


Figure 1. Main window of MagSaki.
Table 1. Magnetic parameters used in MagSaki version 1.0, 2001.
| Symbol | Meaning |
|---|---|
| D | Axial zero-field splitting parameter |
| g | g-factor |
| J | Exchange coupling parameter |
| T | Temperature |
| TIP | Temperature-independent paramagnetism |
| v | v = D/(k l)* |
| D | Axial splitting parameter |
| k | Orbital reduction factor containing the admixture |
| l | Spin-orbit coupling parameter |
| meff | Effective magnetic moment per atom |
| cA | Atomic magnetic susceptibility |
 Return
Return